Org Lett. 2018 Jun 1;20(11):3250-3254. doi: 10.1021/acs.orglett.8b01112. Epub 2018 May 15.
Ben Haj Salah K, Legrand B, Bibian M, Wenger E, Fehrentz JA, Denoyelle S.
Abstract
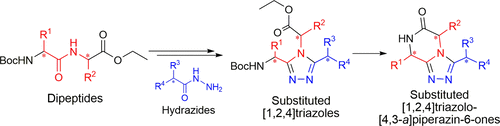
A stereoconservative synthesis to access the triazole-fused ketopiperazine (TKP) scaffold is presented. This underexplored platform offers a wide range of structural modulations with several points of diversity and chiral centers. A series of [1,2,4]triazolo[4,3- a]piperazin-6-ones was synthesized from optically pure dipeptides. The methodology was then successfully applied to access the pyrrolo[1,2- a]triazolo[3,4- c]piperazin-6-one tricycle. Importantly, the crystal structures of representative TKPs confirmed that the configuration of the chiral centers was controlled during the synthetic route and facilitated description of the orientation of the substituents depending on their nature and position on the TKP scaffold.