Chemistry. 2018 Dec 12. doi: 10.1002/chem.201804404. Epub 2018 Nov 15
Simon M, Milbeo P, Liu H, André C, Wenger E, Martinez J, Amblard M, Aubert E, Legrand B, Calmès M.
Abstract
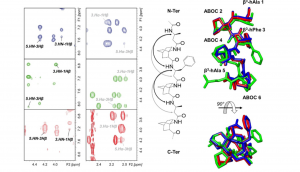
12/10-Helices constitute suitable templates that can be used to design original structures. Nevertheless, they often suffer from a weak stability in polar solvents because they exhibit a mixed hydrogen-bond network resulting in a small macrodipole. In this work, stable and functionalizable 12/10-helices were developed by alternating a highly constrained β2, 3, 3 -trisubstituted bicyclic amino acid (S)-1-aminobicyclo[2.2.2]octane-2-carboxylic acid ((S)-ABOC) and an acyclic substituted β-homologated proteinogenic amino acid (l-β3 -hAA). Based on NMR spectroscopic analysis, it was shown that such mixed β-peptides display well-defined right-handed 12/10-helices in polar, apolar, and chaotropic solvents; that are, CD3 OH, CDCl3 , and [D6 ]DMSO, respectively. The stability of the hydrogen bonds forming the C10 and C12 pseudocycles as well as the benefit provided by the use of the constrained bicyclic ABOC versus typical acyclic β-amino acids sequences when designing 12/10-helix were investigated using NH/ND NMR exchange experiments and DFT calculations in various solvents. These studies showed that the β3 -hAA/(S)-ABOC helix displayed a more stable hydrogen-bond network through specific stabilization of the C10 pseudocycles involving the bridgehead NH of the ABOC bicyclic scaffold.

