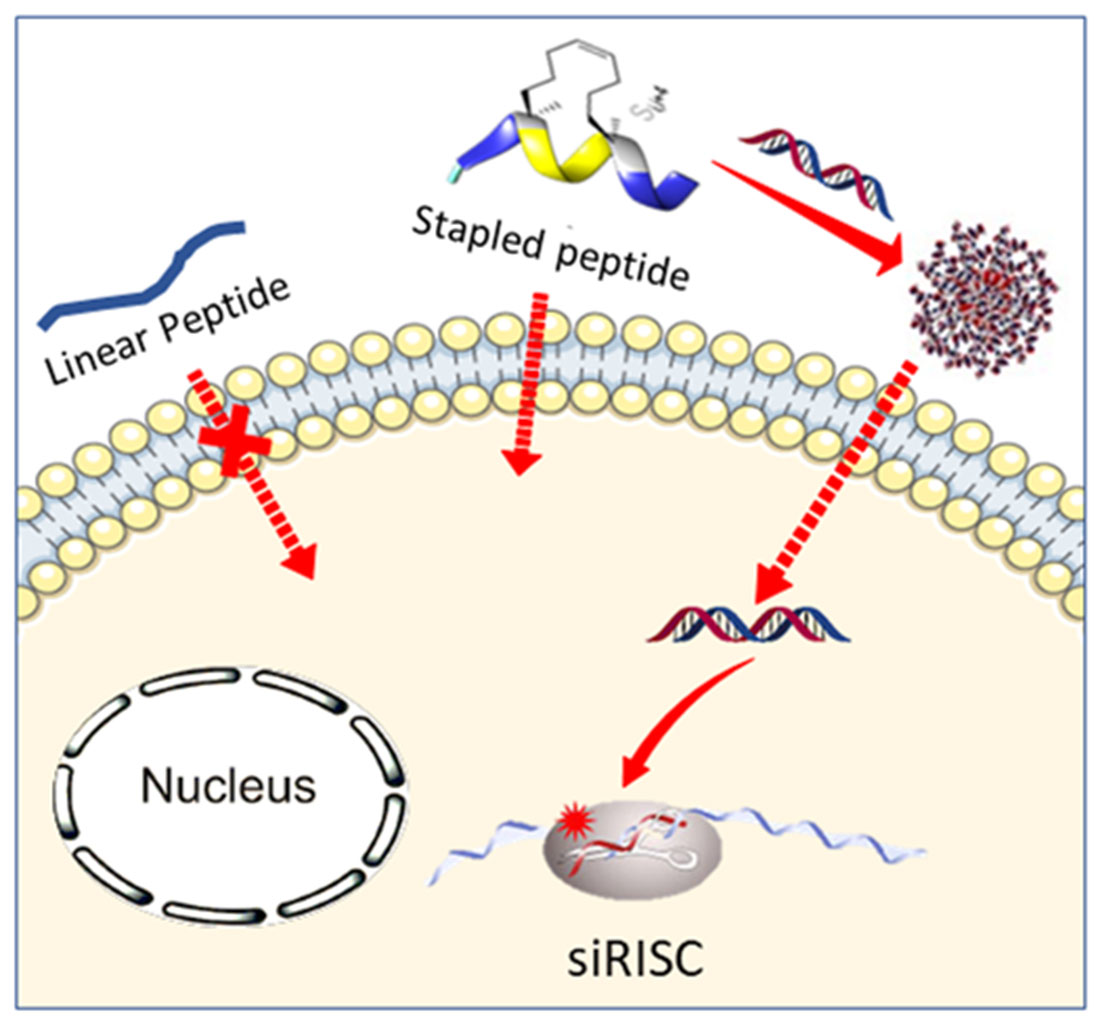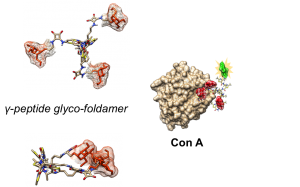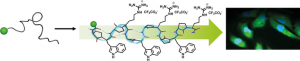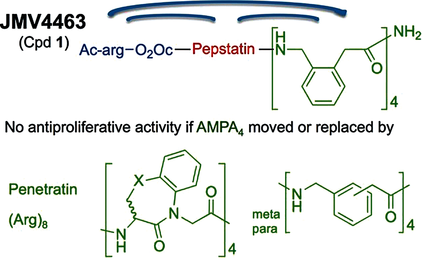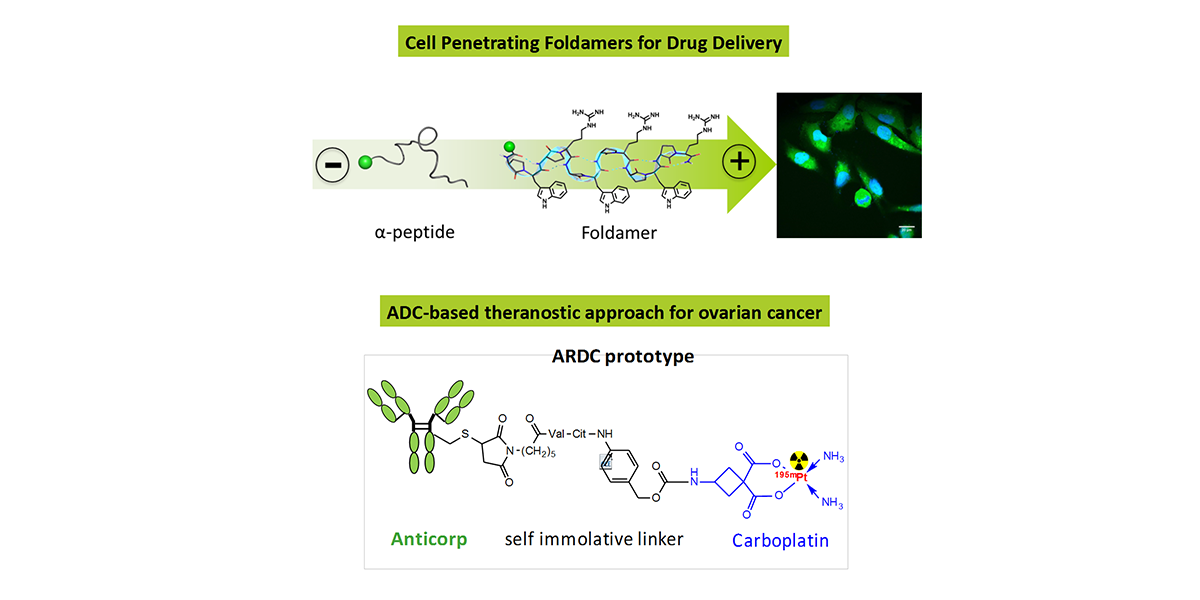
A lot of potential drugs are ineffective because of their inability to cross certain biological membranes, such as the lipid bilayer or the blood brain barrier. Once inside the cell, those compounds often have to find a local address, also known as cell compartment, but unfortunately often they will lose their way and find themselves trapped in the ‘’wrong neighborhoods’’ (cell organelles). In the same time most of the anti-cancer drugs have devastating side effects due to the fact that they target equally healthy and tumour cell. For all those reasons the terms – ‘’Vectorization, targeting and drug delivery’’ are key words in modern medicinal chemistry. In our group we actively pursue the development of cell penetrating and cell targeting compounds based on peptides, foldamers and anti-bodies. These vectors are used to send bioactive compounds, such as anti-cancer drugs, to a preferred cellular compartment or to target a certain organ or cell type in the living species.