Abstract
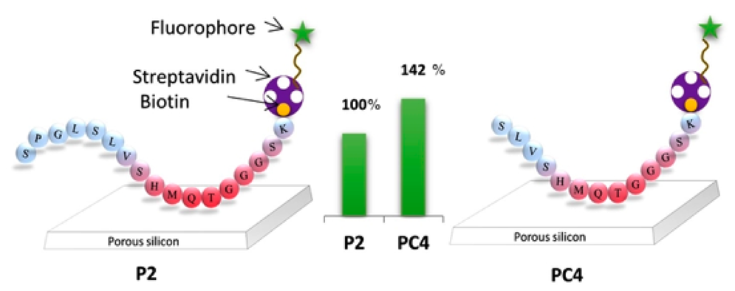
Engineering peptides that present selective recognition and high affinity for a material is a major challenge for assembly-driven elaboration of complex systems with wide applications in the field of biomaterials, hard-tissue regeneration, and functional materials for therapeutics. Peptide-material interactions are of vital importance in natural processes but less exploited for the design of novel systems for practical applications because of our poor understanding of mechanisms underlying these interactions. Here, we present an approach based on the synthesis of several truncated peptides issued from a silicon-specific peptide recovered via phage display technol. We use the photonic response provided by porous silicon microcavities to evaluate the binding efficiency of 14 different peptide derivs. We identify and engineer a short peptide sequence (SLVSHMQT), revealing the highest affinity for p+-Si. The mol. recognition behavior of the obtained peptide fragment can be revealed through mutations allowing identification of the preferential affinity of certain amino acids toward silicon. These results constitute an advance in both the engineering of peptides that reveal recognition properties for silicon and the understanding of biomol.-material interactions.