Abstract
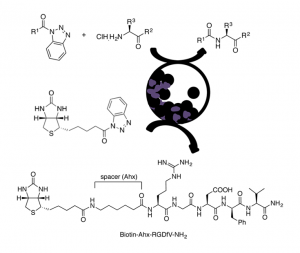
An eco-friendly methodol. for prepg. Fmoc-, Z-, and Boc-N-protected dipeptides and tripeptides is described, from the corresponding N-protected-α-aminoacyl benzotriazoles and α-amino acid derivs., with different C-terminal functionalities such as esters or amides, using vibrational ball-mill (VBM). The reactivity of a β-amino ester was also investigated. In some cases, the coupling was achieved by liq.-assisted grinding (LAG). α,α- and one α,β-dipeptide were obtained in good to excellent yields mainly by pptn. in water, resulting in an improved environmental impact compared to classical peptide synthesis in soln., as shown by green metric calcns. The method was extended to the biotinylation, via an aminohexanoyl spacer, of the pentapeptide RGDfV, which contains the well-known integrins recognition site arginine-glycine-aspartic acid (RGD) motif.