Abstract
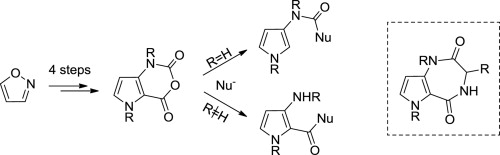
A convenient synthesis of pyrrolo[3,2-d][1,3]oxazine-2,4-dione is described and its reactivity towards various nucleophiles studied. The regioselective ring opening of pyrrolo[3,2-d][1,3]oxazine-2,4-dione or its N-alkylated analog in the presence of alanine or proline afforded, resp., imidazolidinedione and 2 N-protected pyrrolo[3,2-e][1,4]diazepines in a one-pot process. In a last part of this study, an alternative route to produce a library of eight non protected pyrrolo[3,2-e][1,4]diazepine-2,5-diones is described to overcome the limited reactivity of pyrrolo[3,2-d][1,3]oxazine-2,4-dione.