Eur. J. Med. Chem., 2022, 241, 114619, https://doi.org/10.1016/j.ejmech.2022.114619
R. Mustière, P. Lagardère, S. Hutter, V. Dell’Orco, N. Amanzougaghene, S. Tajeri, J.-F. Franetich, S. Corvaisier, Marc Since, A. Malzert-Fréon, N. Masurier, V. Lisowski, P. Verhaeghe, D. Mazier, N. Azas, P. Vanelle, N. Primas
Abstract
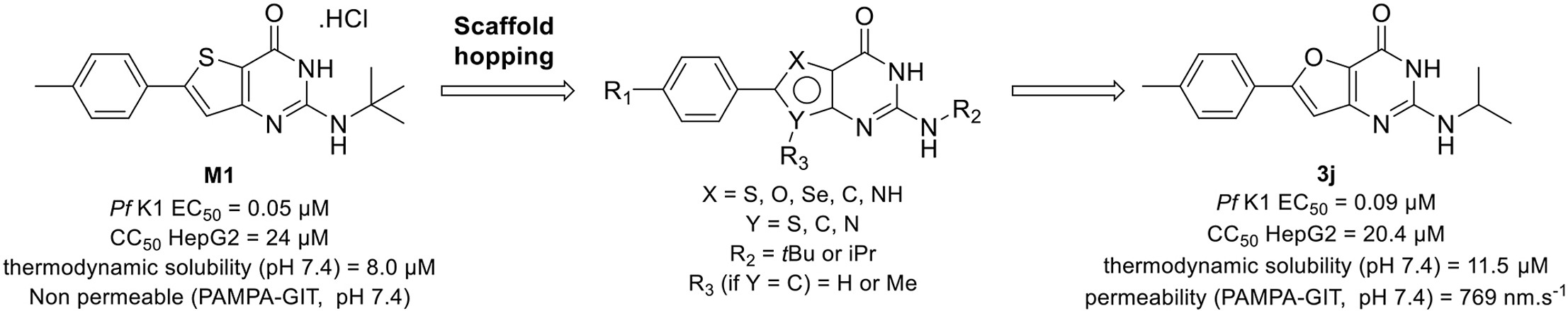
Gamhepathiopine (also known as M1), is a multi-stage acting antiplasmodial 2-tert-butylaminothieno[3,2-d]pyrimidin-4(3H)-one hydrochloride that was first described in 2015. The development of this compound is limited by poor microsomal stability, insufficient aqueous solubility and low intestinal permeability. In order to obtain new optimized derivatives, we conducted a scaffold hopping strategy from compound M1, resulting in the synthesis of 20 new compounds belonging to six chemical series. All the compounds were tested on the K1 multi-resistant strain of Plasmodium falciparum and the human HepG2 cell-line, to evaluate their antiplasmodial activity and their cytotoxicity. Analogues’ biological results also highlighted the mandatory presence of a heteroatom at position 5 of the thieno[3,2-d]pyrimidin-4(3H)-one moeity for the antiplasmodial activity. However, modifications at position 7 were detrimental for the antiplasmodial activity. We identified furane bioisostere 3j as a promising candidate, showing good blood stage antiplasmodial activity, better water solubility and highly improved intestinal permeability in the PAMPA assay.

